Summary
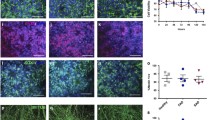
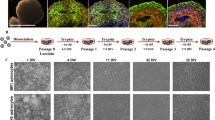
Primary cultures of the brain from sheep embryos were used to establish cell lines after transfection by the simian virus 40 (SV40) large T gene. Two of the lines (A15 and 4A6) displayed astroglial properties. They expressed the glial fibrillary acidic protein (GFAP), intermediate filament protein vimentin, and S-100 (beta-subunit) protein. While numerous rodent and human glial cell lines are available, this is to our knowledge the first description of ovine cell lines with astrocyte features. In addition, these cell lines were derived from sheep embryos chosen for their genetic susceptibility to scrapic (PrP genotype: VV136, QQ171). Therefore, they could be attractive tissue culture models for the study of propagation and pathogenesis of the scrapie agent ex vivo.
Similar content being viewed by others
References
Anderson, C. A.; Higgins, R. J.; Waldvogel, A. S.; Osburn, B. I. Tropism of border disease virus for oligodendrocytes in ovine fetal brain cellcultures. Am. J. Vet. Res. 48:822–827; 1987.
Bigami, A.; Dahl, D.; Gliosis. In: Kettenmann, H.; Ransom, B. R., ed.. Neuroglia. Oxford: Oxford University Press; 1995:843–858.
Brandner, S.; Isenmann, S.; Kühne, G.; Aguzzi, A. Identification of the end stage of scrapie using infected neural grafts. Brain Pathol. 8:19–27; 1998.
Brandner, S.; Isenmann, S.; Raeber, A.; Fisher, M.; Sailer, A.; Kobayashi, Y.; Marino, S.; Weissmann, C.; Aguzzi, A. Normal host prion protein necessary for scrapie-induced neurotoxicity. Nature, 379:339–343; 1996.
Büeler, H.; Aguzzi, A.; Sailer, A.; Greiner, R. A.; Autenried, P.; Aguet, M. Weissmann, C. Mice devoid of PrP are resistant to scrapie. Cell 73: 1339–1347; 1993.
Clouscard, C.; Beaudry, P.; Elsen, J. M.; Milan, D.; Dussaucy, M.; Bounneau, C Schelchert, F.; Chatelain, J.; Launay, J. M.; Laplanche, J. L. Different allelic effects of the codons 136 and 171 of the prion protein gene in sheep with natural scrapie. J. Gen. Virol. 76:2097–2101; 1995.
Diedrich, J. F.; Bendheim, P. E.; Kim, Y. S.; Carp, R. I.; Haase, A. T. Scrapieassociated prion protein accumulates in astrocytes during scrapie infection. Proc. Natl. Acad. Sci. USA 88:375–379; 1991.
Eklund, C. M.; Kennedy, R. C.; Hadlow, W. J. Pathogenesis of scrapie virus infection in the mouse J. Infect. Dis. 117:15–22; 1967.
Elder, G. A.; Potts, B. J.; Sawyer, M. Characterization of glial subpopulations in cultures of the ovine central nervous system Glia. 1:317–327; 1988.
Eng, L. F. Glial fibrillary acidic protein (GFAP): the major protein of glial intermediate filaments in differentiated astrocytes J. Neuroimmunol. 8:203–214; 1985.
Eng, L. F.; Lee, Y. E. Intermediate filaments in astrocytes. In: Kettenmann, H.; Ransom, B. R., ed. Neuroglia. Oxford: Oxford University Press; 1995:650–667.
Evrard, C.; Galiana, E. Rouget, P. Establishment of “normal” nervous cell lines after transfert of polyoma virus and adenovirus early genes into murine brain cells EMBO J. 5:3157–3162; 1986.
Fraser, H.; Dickinson, A. G. Pathogenesis of scrapie in the mouse: the role of the spleen Nature. 226:462–463; 1970.
Fedoroff, S.; White, R.; Subrahmanyan, L.; Kalnins, V. I. Astrocyte cell lineage II. mouse fibrous astrocytes and reactive astrocytes in culture have vimentin-and GFP-containing intermediate filaments Brain Res. 283:303–315; 1983.
Galiana, E.; Borde, I.; Marin, P.; Rassoulzadegan, M.; Cuzin, F.; Gros, F.; Rouget, P.; Evrard, C. Establishment of permanent astroglial cell lines, able to differentiate ex vivo, from transgenic mice carrying the polyomavirus large T gene: an alternative approach to brain cell immortalization. J. Neurosci. Res. 26:269–277; 1990.
Ghandour, M. S.; Labourdette, G.; Vincendon, G.; Gombos, G.. A biochemical and immunohistological study of S100 protein in developing rat cerebellum. Dev. Neurosci. 4:98–109; 1981.
Gown, A. M.; Vogel, A. M. Monoclonal antibodies to intermediate filaments of human cells—unique and cross-reacting antibodies J. Cell Biol. 95:414–424; 1982.
Harlow, E.; Crawford, L. V.; Pim, D. C.; Williamson, N. M. Monoclonal antibodies specific for simian virus 40 tumor antigens J. Virol. 39:861–869; 1981.
Hunter, N. PrP genetics in sheep and the implications for scrapie and BSE. Trends Microbiol. 5:331–334; 1997.
Ludwin, S. K.; Szuchet, S. Myelination by mature oligodendrocytes in vivo and in vitro: evidence that different steps in the myelination process are independently controlled Glia 8:219–231; 1993.
McCarthy, K. D.; de Vellis, J. Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue J. Cell Biol 85: 890–902; 1980.
Markovits, P.; Dormont, D.; Delpech, B.; Court, L.; Latarjet, R. Trials of ex vivo propagation of the scrapie agent in mouse nerve cells C. R. Acad. Sci. Paris 293:413–417; 1981.
Moser, M.; Collelo, R. J.; Pott, U.; Oesch, B. Developmental expression of the prion protein gene in glial cells Neuron 14:509–517; 1995.
Oblinger M. M.; Kost, S. A.; Singh, L. D. In: Federoff, S.; Juurlink, B. M. J.; Doucette, R., ed. Biology and pathology of astrocyte-neuron interactions. Plenum Press, New York: 1993:291–302.
Prusiner, S. B. Molecular biology and pathogenesis of prion diseases Trends Biochem. Sci. 21:482–487; 1996.
Quinlan, R. A.; Franke, W. W. Molecular interactions in intermediate-sized filaments revealed by chemical crosslinking: heteropolymers of vimentin and glial fibrillary acidic protein in cultured human glioma cells Eur. J. Biochem. 132:477–484; 1983.
Race, R. The scrapie agent ex vivo Curr. Top. Microbiol. Immunol. 172:181–193; 1991.
Raeber, A. J.; Race, R. E.; brandner, S.; Priola, S. A.; Sailer, A.; Bessen, R. A.; Mucke, L.; Manson, J.; Aguzzi, A.; Oldstone, M. B. A.; Weissmann, C.; Chesebro, B. Astrocyte-specific expression of hamster prion protein (PrP) renders PrP knockout mice susceptible to hamster scrapie, EMBO J. 16:6057–6065; 1997.
Richard, O.; Duittoz, A. H.; Hevor, T. K. Early, middle, and late stages of neural cells from ovine embryo in primary cultures Neurosci. Res. 31:61–68; 1998.
Sailer, A.; Büeler, H.; Fisher, M.; Aguzzi, A.; Weissmann, C. No propagation of prions in mice devoid of PrP Cell 77:967–968; 1994.
Schuermann, M. An expression vector for stable expression of oncogenes. Nucleic Acids Res. 18:4945–4946; 1990.
Schwartz, B.; Vicard, P.; Delouis, C.; Paulin, D. Mammalian cell lines can be efficiently established ex vivo upon expression of the SV40 large T antigen driven by a promoter sequence derived from the human vimentin gene Biol. Cell. 73:7–14; 1991.
Sharp, G.; Osborn, M.; Weber, K. Occurrence of two different intermediate filament proteins in the same filament in situ within a human glioma cell line. An immunoelectron microscopical study Exp. Cell Res. 141:385–395; 1982.
Wang, F.; Cairncross, J. G.; Liem, R. K. H. Identification of glial filament protein and vimentin in the same intermediate filament system in human glioma cells Proc. Natl. Acad. Sci. USA 81:2102–2106; 1984.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vilette, D., Madelaine, M.F. & Laude, H. Establishment of astrocyte cell lines from sheep genetically susceptible to scrapie. In Vitro Cell.Dev.Biol.-Animal 36, 45–49 (2000). https://doi.org/10.1290/1071-2690(2000)036<0045:EOACLF>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2000)036<0045:EOACLF>2.0.CO;2